Records & Data Management
Our Services
Document Storage
Digital Information Management / Scanning
Our document management system for hard-copy documents, files and cartons provides convenient access to information stored within your records system.
File Management
In-house Records Web Portal
Multi Media Management
Like the money you save regularly, sending data off-site is an insurance against unpredictable events.
Secure Destruction / Shredding
Workplace Relocation
Moving needn’t be a big distraction. Highly trained and well organised, our moving teams bring efficiency to every relocation.
Document Storage
File Management
Digital Information Management / Scanning
Multi Media Management
In-house Records Web Portal
Secure Destruction / Shredding
Workplace Relocation
Multi Media Management
In-house Records Web Portal
Workplace Relocation
Secure Destruction / Shredding
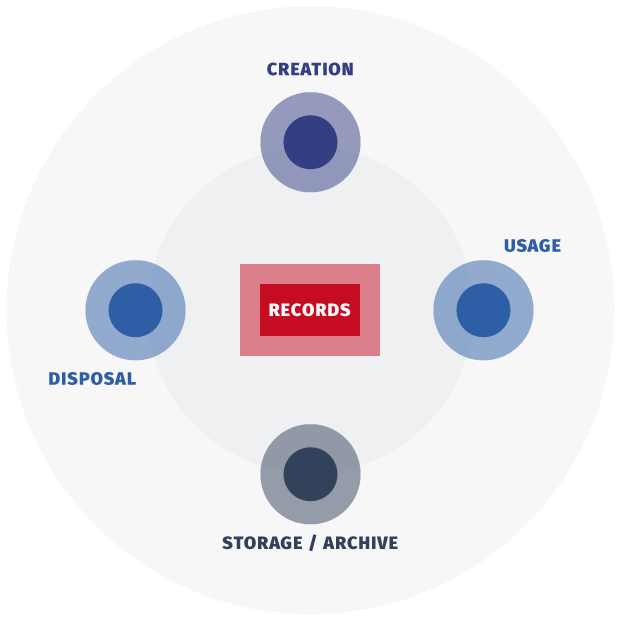
Records and Data Lifecycle
1. Different type of people – Our experience, knowledge, and nthusiasm give you new perspectives.
2. World-class technology – We continuously develop technology’s potential using market and customer insights.
3. Quality attitude – Performance, compliance and confidence are the customer outcomes we seek.
4. Serious about security – For the most confidential and the highest level of protection, our security procedures go beyond the industry standard.
certifications
- ISO 9001:2015 Quality Management.
- ISO 14001:2015 Environmental Management System.
- OHSAS 18001:2007 Health & Safety Management System.
- ISO 22301:2012 Business Continuity.
- ISO 27001 Information Security Management.













